Now Reading: Is Ozone A Reliable Proxy For Molecular Oxygen? III. The Impact Of CH4 On The O2-O3 Relationship For Earth-like Atmospheres
-
01
Is Ozone A Reliable Proxy For Molecular Oxygen? III. The Impact Of CH4 On The O2-O3 Relationship For Earth-like Atmospheres
Is Ozone A Reliable Proxy For Molecular Oxygen? III. The Impact Of CH4 On The O2-O3 Relationship For Earth-like Atmospheres
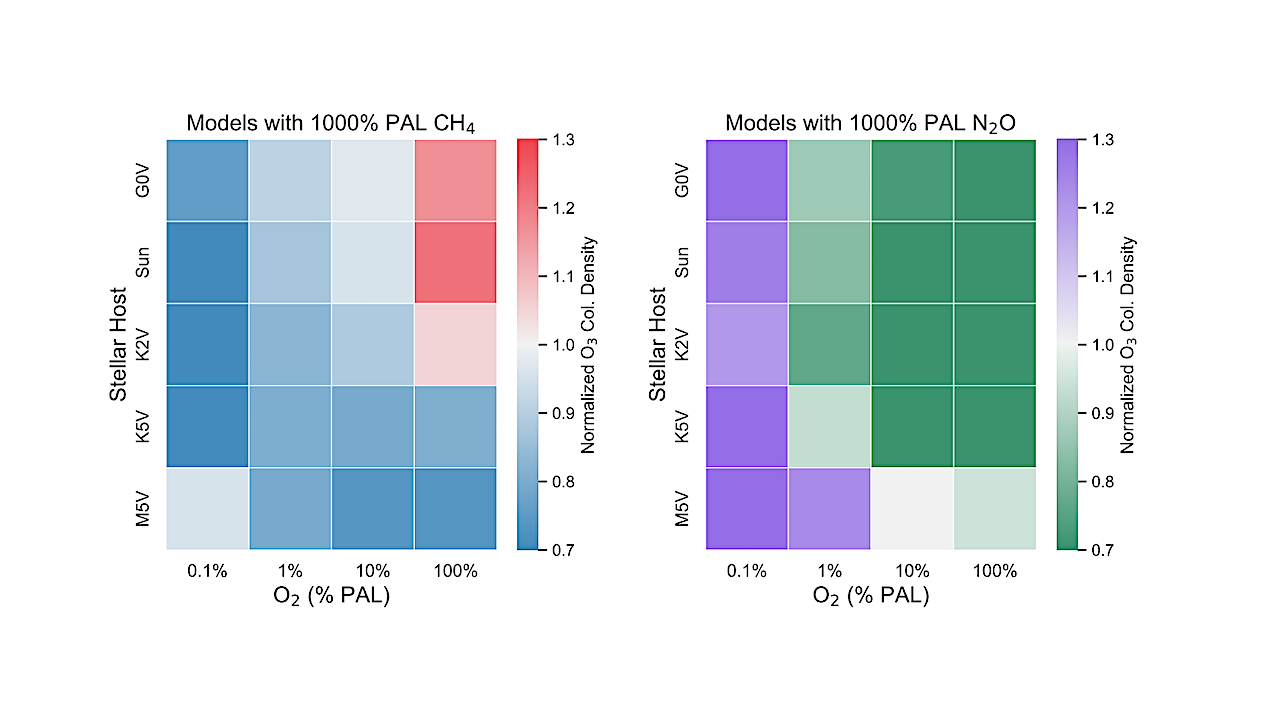
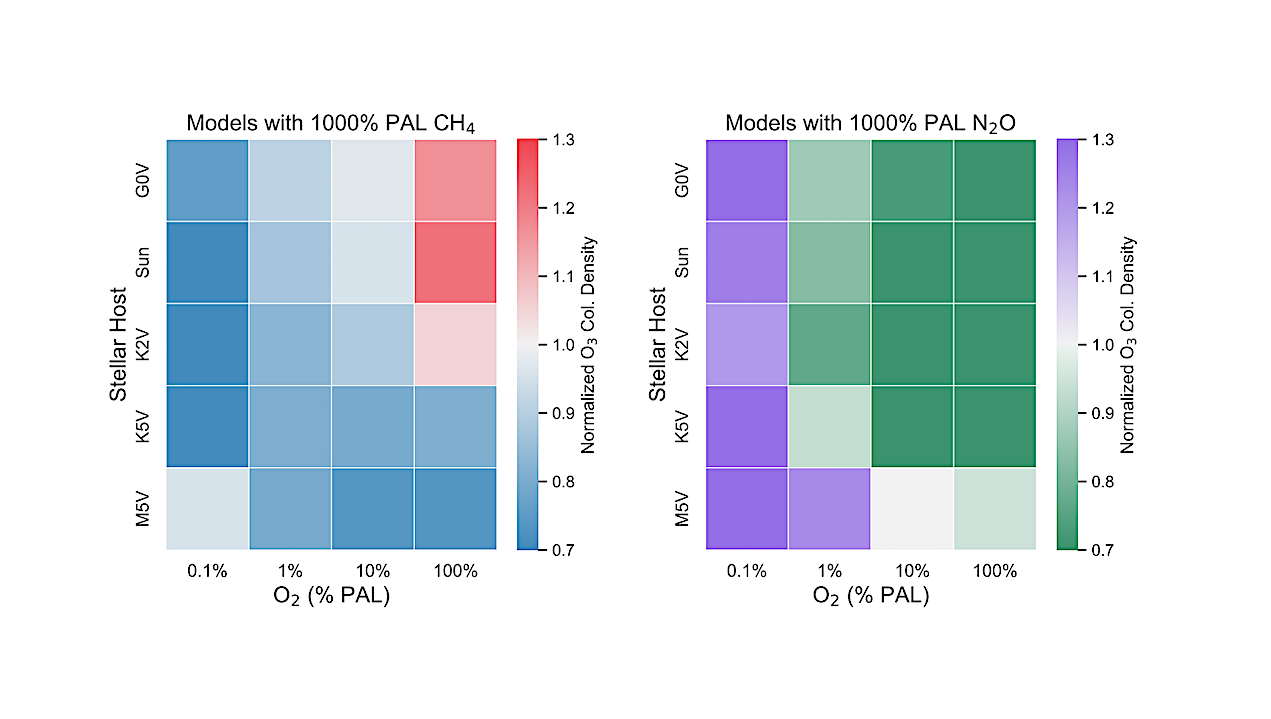
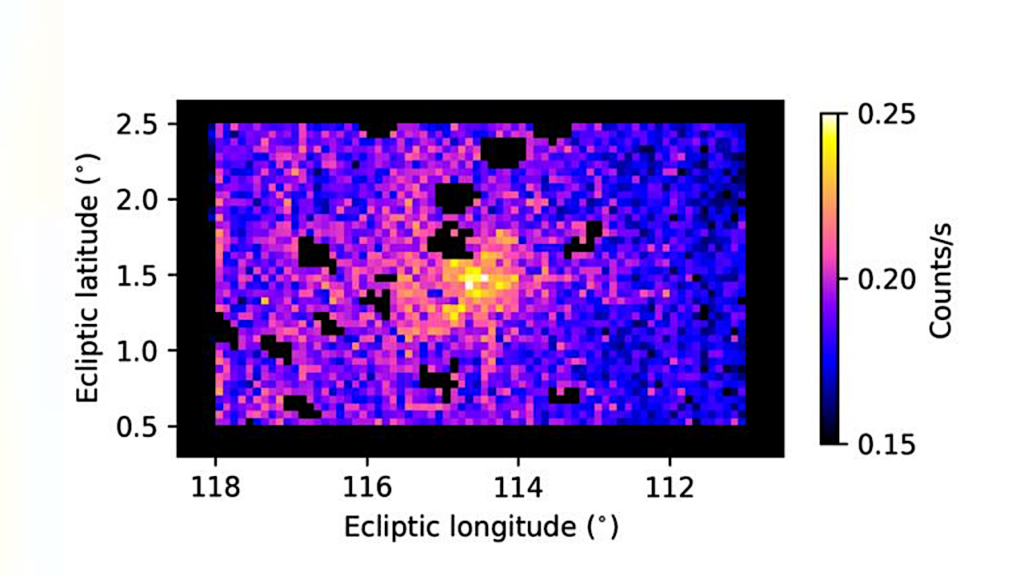
Abundances of O3 for both high CH4 models from this study and high N2O models from Kozakis et al. (2025) normalized to the amount of O3 with modern amounts of CH4 and N2O for all host stars at 0.1%, 1%, 10%, and 100% PAL O2. Both figures share the same color bar limits in order to facilitate comparisons. — astro-ph.EP
In the search for life in the Universe, molecular oxygen (O2) combined with a reducing species, such as methane (CH4), is considered a promising disequilibrium biosignature.
In cases where it would be difficult or impossible to detect O2 (e.g., mid-IR or low O2 levels), it has been suggested that ozone (O3), the photochemical product of O2, could be used as a proxy for determining the abundance of O2. As the O2-O3 relationship is nonlinear, the goal of this series of papers is to explore how it would change for different host stars and atmospheric compositions and learning how to use O3 to infer O2.
We used photochemistry and climate modeling to further explore the O2-O3 relationship by modeling Earth-like planets with the present atmospheric level (PAL) of O2 between 0.01% to 150% along with high and low CH4 abundances of 1000% and 10% PAL, respectively. Methane is of interest not only because it is a biosignature, but also the source of hydrogen atoms for hydrogen oxide (HOx) which destroys O3 through catalytic cycles and acts as a catalyst for the smog mechanism of O3 formation in the lower atmosphere.
We find varying CH4 causes changes to the O2-O3 relationship in ways that are highly dependent on both host star and O2 abundance. A striking result for high CH4 models in high O2 atmospheres around hotter hosts is that enough CH4 is efficiently converted into H2O to significantly impact stratospheric temperatures, and therefore the formation/destruction rates of O3.
Changes in HOx also influenced the HOx catalytic cycle and smog O3, causing variations in harmful UV reaching the surface as well as changes in the 9.6~μm O3 feature in emission spectra. This demonstrates the need to explore the O2-O3 relationship in order to use O3 as a reliable proxy for O2 in future observations.
Thea Kozakis, João M. Mendonça, Lars A. Buchhave, Luisa M. Lara
Comments: In production with A&A
Subjects: Earth and Planetary Astrophysics (astro-ph.EP)
Cite as: arXiv:2508.19062 [astro-ph.EP] (or arXiv:2508.19062v1 [astro-ph.EP] for this version)
https://doi.org/10.48550/arXiv.2508.19062
Focus to learn more
Related DOI:
https://doi.org/10.1051/0004-6361/202556015
Focus to learn more
Submission history
From: Thea Kozakis
[v1] Tue, 26 Aug 2025 14:21:28 UTC (682 KB)
https://arxiv.org/abs/2508.19062
Astrobiology
Stay Informed With the Latest & Most Important News
-
 01Two Black Holes Observed Circling Each Other for the First Time
01Two Black Holes Observed Circling Each Other for the First Time -
 02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life
02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life -
 03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series)
03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series) -
 04True Anomaly hires former York Space executive as chief operating officer
04True Anomaly hires former York Space executive as chief operating officer -
 05Φsat-2 begins science phase for AI Earth images
05Φsat-2 begins science phase for AI Earth images -
 06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters
06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters -
 07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly
07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly