Now Reading: Can Cyanide Radicals Drive Molecular Backbone Growth On Interstellar Icy Grains?
-
01
Can Cyanide Radicals Drive Molecular Backbone Growth On Interstellar Icy Grains?
Can Cyanide Radicals Drive Molecular Backbone Growth On Interstellar Icy Grains?
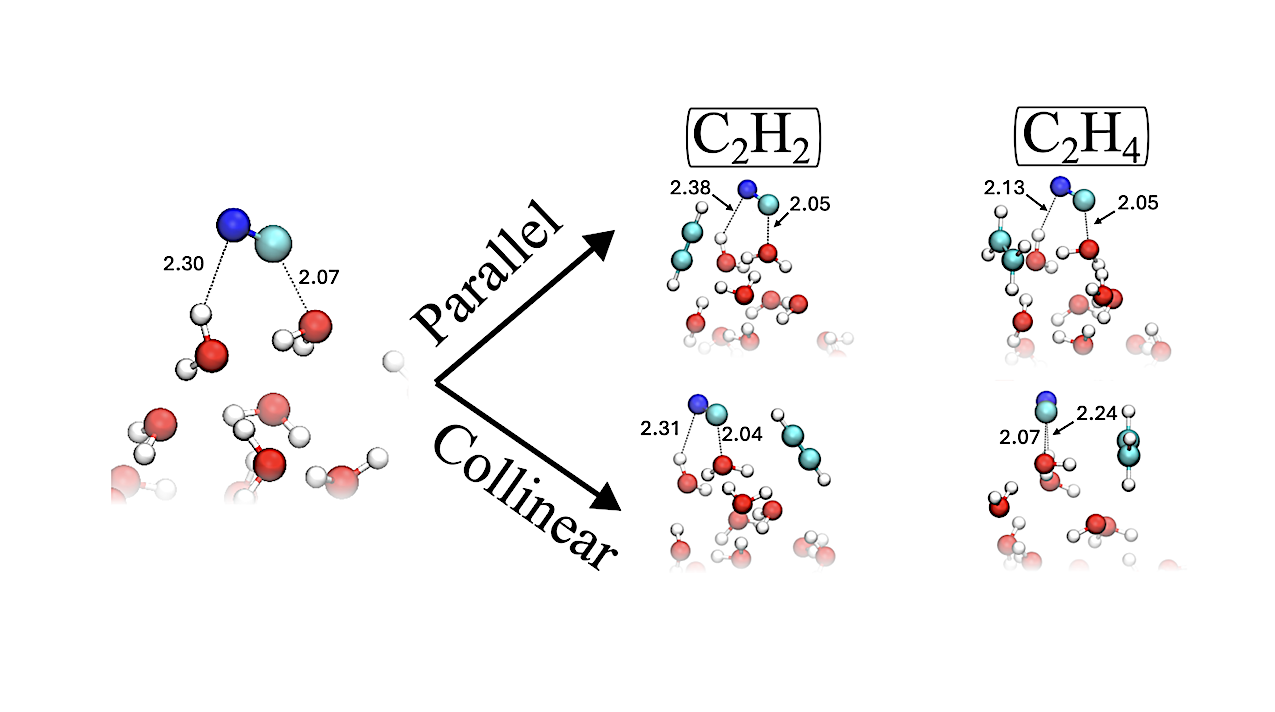

(Left) Depiction of the hemibonded CN radical adsorbed on a 14 H2O cluster. (Right, Top) Parallel geometries for the approach of C2H2 and C2H4 to hemibonded CN. (Right, Bottom) Collinear geometries for the approach of C22H2 and C2H4 to hemibonded CN. All the structures are optimized and represent the actual reactant states used in the determination of the energetic descriptors of the reaction. — astro-ph.GA
Motivated by the value of CN-bearing molecules as tracers of interstellar physical conditions, we investigate the reactions of adsorbed CN radicals with acetylene and ethylene (C2H2 and C2H4) on interstellar dust-grain analogues using quantum chemical calculations.
We find that reactivity is strongly controlled by the relative orientation of the reactants, with specific geometries either promoting or inhibiting reaction. We further show that, on ice, these reactions differ qualitatively from their gas-phase counterparts, stalling at the formation of the adduct complexes C2H2CN and C2H4CN and exhibiting newly emerged kinetic barriers for the neutral-radical association.
We contextualize our calculations in the same reaction-diffusion framework that would be employed in astrochemical models, finding that, depending on the diffusion energy of the hydrocarbons, these reactions can be either negligible or efficient, highlighting the importance of the local ice structure in interstellar grain chemistry. These findings caution against the use of CN-based tracers that assume barrierless, bimolecular surface reactions involving CN radicals.
Germán Molpeceres, Joan Enrique-Romero
Comments: Accepted for publication in ApJ
Subjects: Astrophysics of Galaxies (astro-ph.GA)
Cite as: arXiv:2603.15138 [astro-ph.GA] (or arXiv:2603.15138v1 [astro-ph.GA] for this version)
https://doi.org/10.48550/arXiv.2603.15138
Focus to learn more
Submission history
From: German Molpeceres
[v1] Mon, 16 Mar 2026 11:34:54 UTC (3,071 KB)
https://arxiv.org/abs/2603.15138
Astrobiology, Astrochemistry,
Stay Informed With the Latest & Most Important News
Previous Post
Next Post
-
 01Two Black Holes Observed Circling Each Other for the First Time
01Two Black Holes Observed Circling Each Other for the First Time -
 02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life
02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life -
 03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series)
03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series) -
 04True Anomaly hires former York Space executive as chief operating officer
04True Anomaly hires former York Space executive as chief operating officer -
 05Φsat-2 begins science phase for AI Earth images
05Φsat-2 begins science phase for AI Earth images -
 06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters
06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters -
 07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly
07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly