Now Reading: Redox Chemistry Of Early Earth And The Origin Of Life
-
01
Redox Chemistry Of Early Earth And The Origin Of Life
Redox Chemistry Of Early Earth And The Origin Of Life
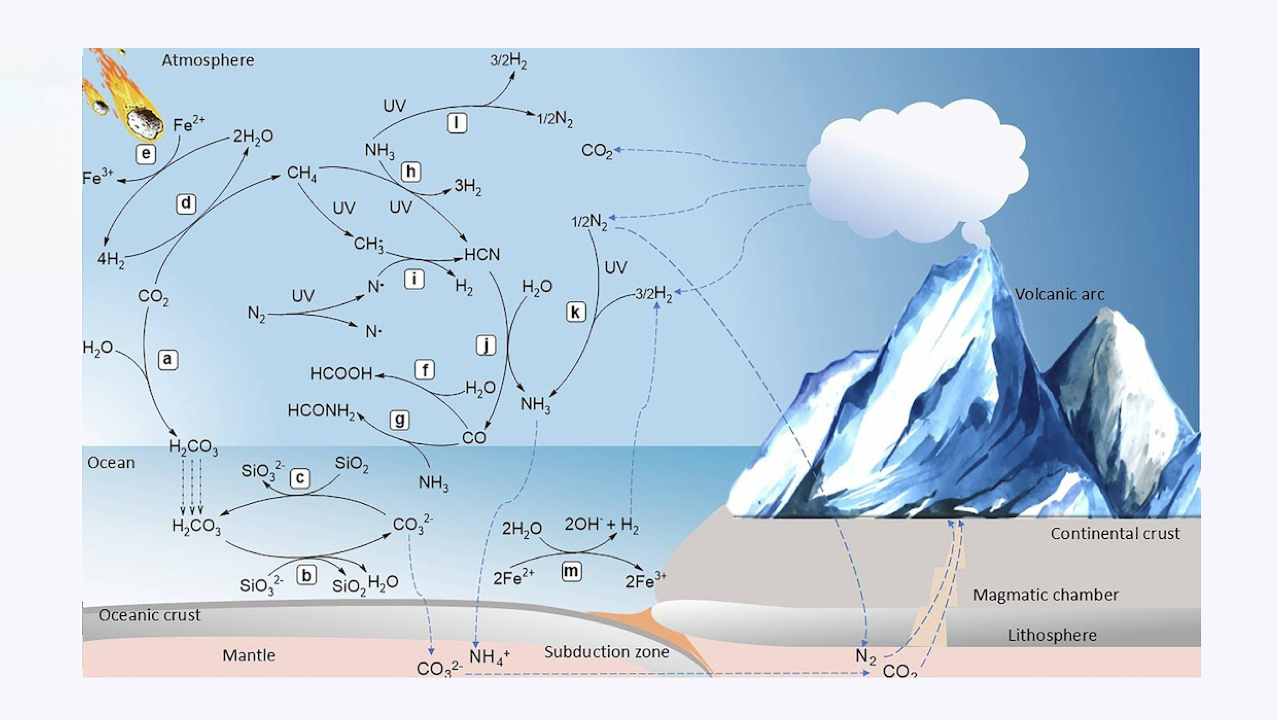
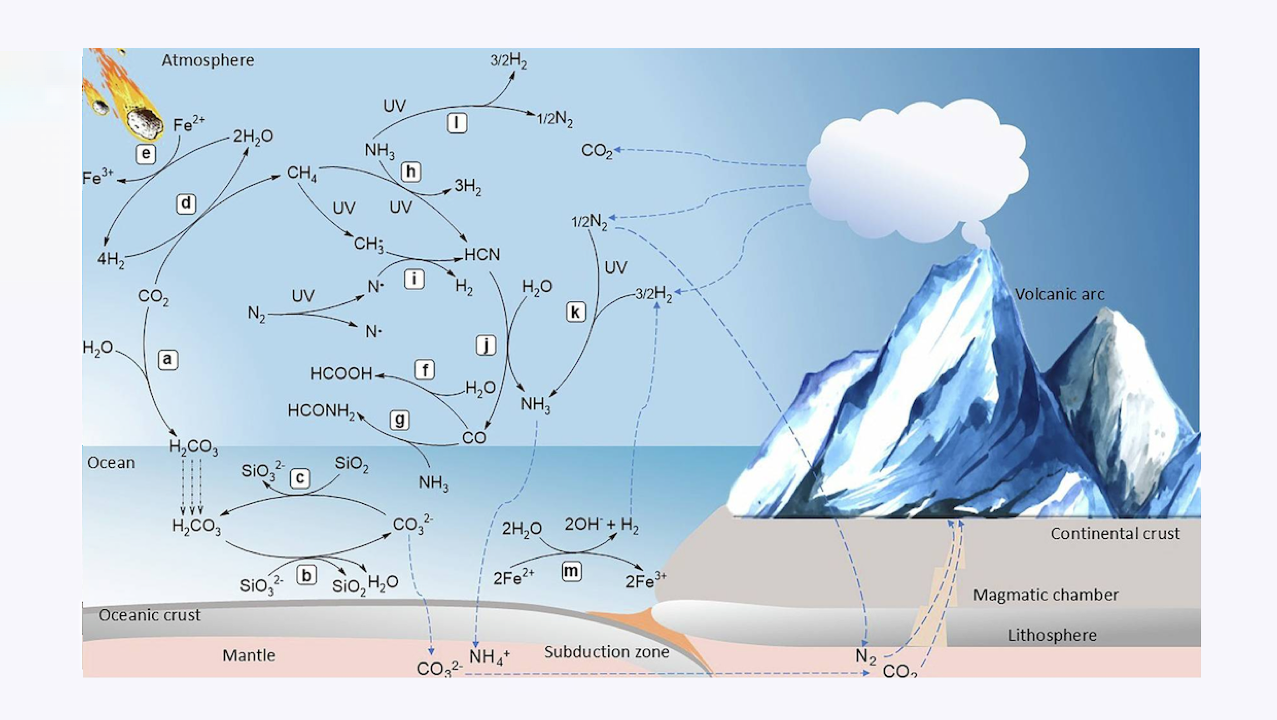
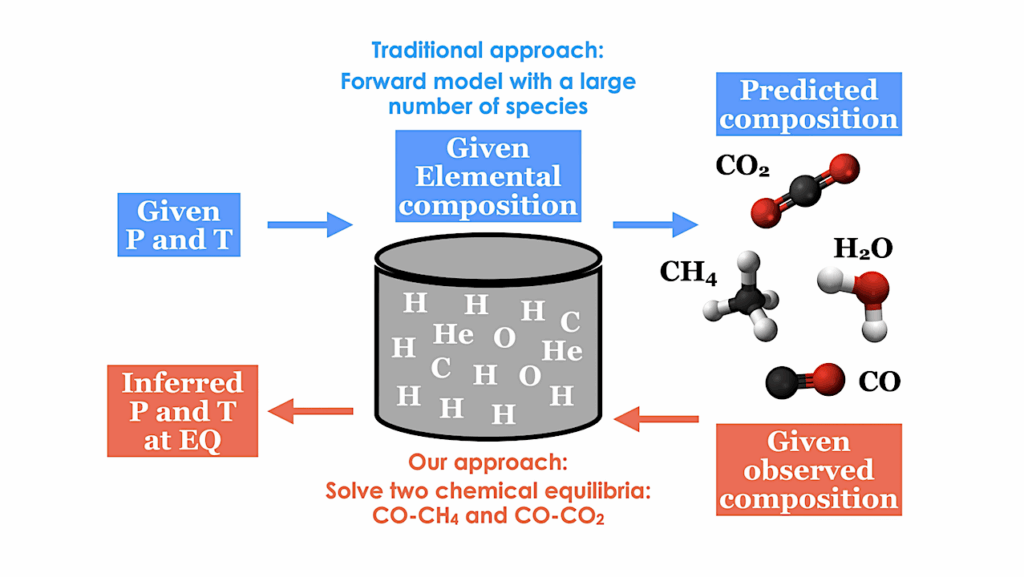
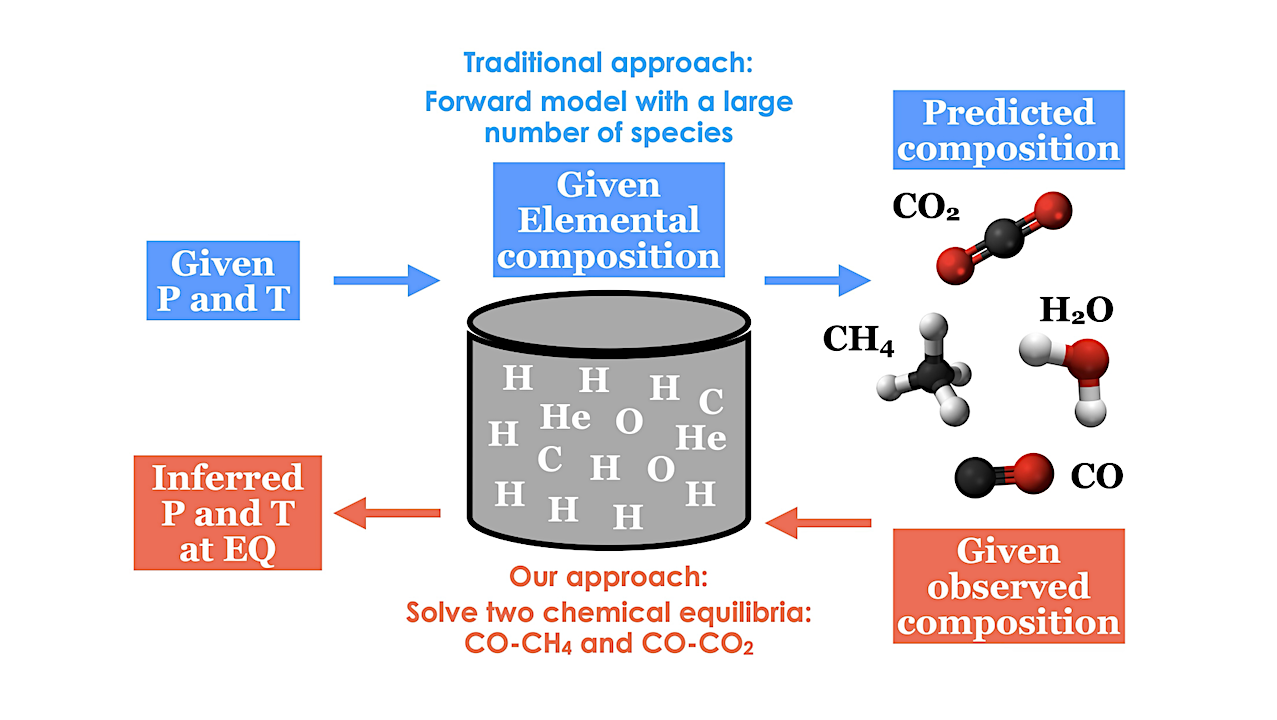
Carbon-silicate cycle includes a carbon dioxide (CO2) dissolution in rainwater to form carbonic acid (H2CO3) that falls on land and oceans; b reaction of H2CO3 and silicate (SiO32-) to form silicon dioxide (SiO2) and carbonate (CO32-), which is sequestered into oceanic crust for further recycling to the mantle followed by release into the atmosphere through volcanic activity; and c regeneration of CO2 through reaction between SiO2 and CO32-. Carbon fixation includes (d) reduction of CO2 to methane (CH4) through (e) the production of H2 from H2O on the surface of iron sulfide minerals that serve as sources of dissolved ferrous ion (Fe2+), which is photochemically oxidized to ferric ions (Fe3+.) in water vapor. Carbon monoxide (CO) either reacts with (f) H2O for synthesis of formic acid (HCOOH) or g ammonia (NH3) for synthesis of formamide (HCONH2). Hydrogen cyanide (HCN) formation may occur through either h reaction between CH4 and NH3 or i reaction of CH3• and N• through free radical mechanism. j HCN further reacts with H2O to produce CO and NH3.that also is produced through (k) N2 fixation under high temperatures, both can be sequestered into the mantle; l NH3 photolysis by UV radiation gives rise to N2 and H2; m hydrothermal H2 production occurs through the H2O oxidation by Fe2+ into H2 used for N2 fixation in the atmosphere. In this figure, royalty-free mountain and meteorite images designed by Freepik platform (www.freepik.com) are used. — Nature via PubMed (open access)
Stay Informed With the Latest & Most Important News
-
 01Two Black Holes Observed Circling Each Other for the First Time
01Two Black Holes Observed Circling Each Other for the First Time -
 02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life
02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life -
 03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series)
03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series) -
 04True Anomaly hires former York Space executive as chief operating officer
04True Anomaly hires former York Space executive as chief operating officer -
 05Φsat-2 begins science phase for AI Earth images
05Φsat-2 begins science phase for AI Earth images -
 06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters
06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters -
 07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly
07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly