Now Reading: The Fate of Frozen Carbonated Water at Europa-like Conditions
-
01
The Fate of Frozen Carbonated Water at Europa-like Conditions
The Fate of Frozen Carbonated Water at Europa-like Conditions
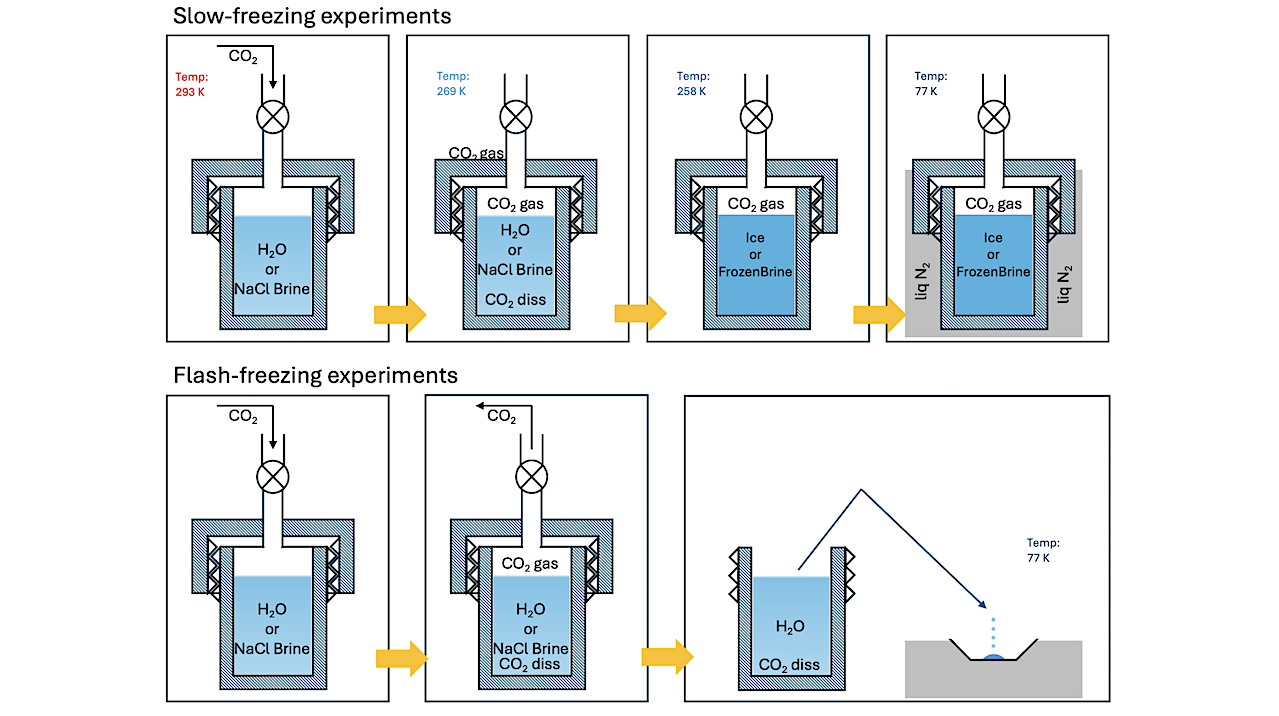
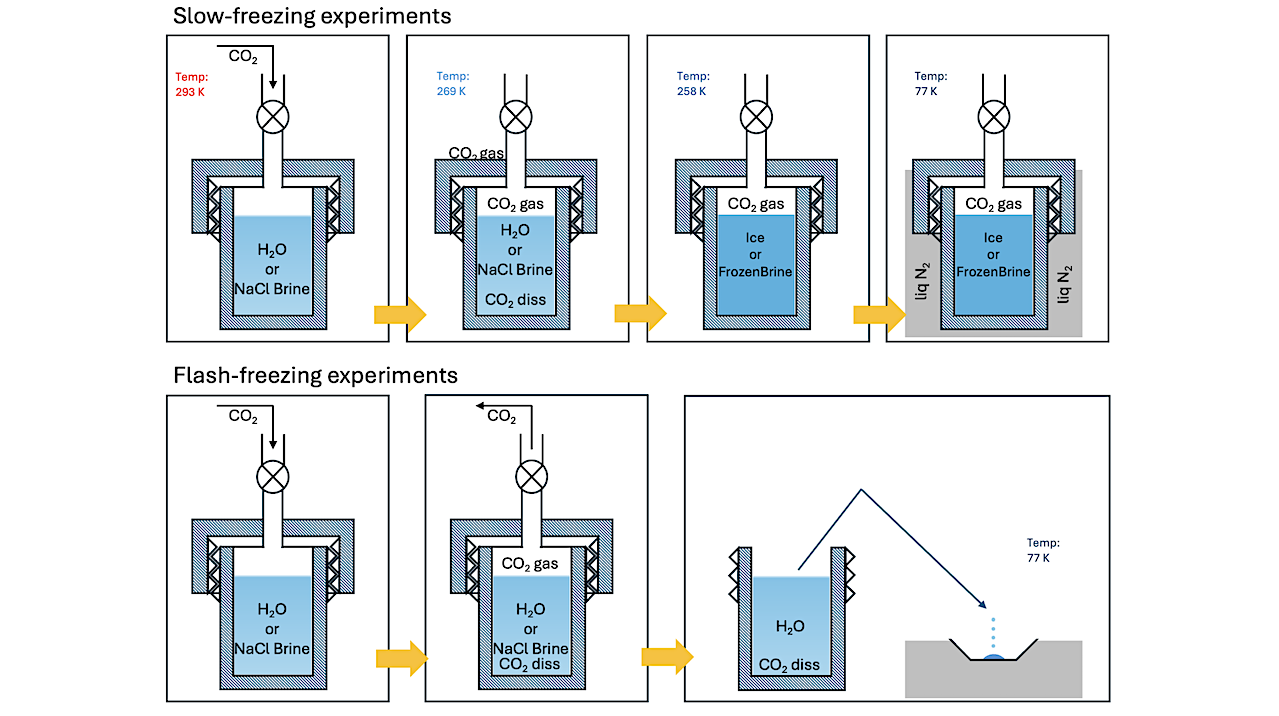
Schematic of the procedure in two sets of experiments performed with carbonated water. The Planetary Science Journal
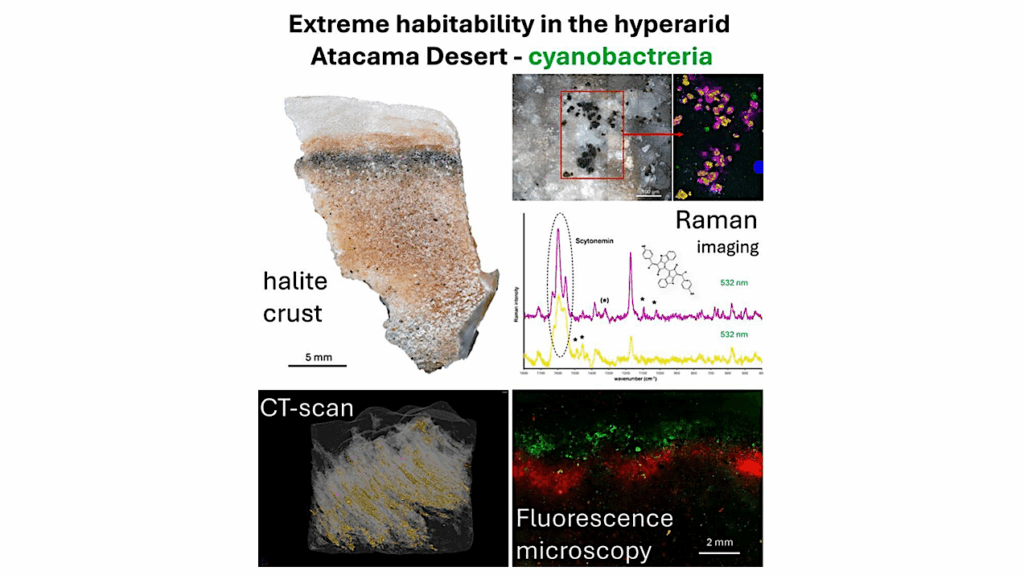
We present the results of experiments probing the retention of CO2 in crystalline water ice, frozen sodium chloride (NaCl) brines, and flash-frozen carbonated water using diffuse reflectance infrared spectroscopy.
Characteristic absorptions alluding to the formation of clathrate hydrates in crystalline ices and frozen brines are observed. NaCl in frozen brines does not qualitatively affect the formation of clathrate hydrates. Generation and stability of clathrates in crystalline ice transiently subjected to pressure–temperature (P–T) conditions in the stability region is observed, despite conditions being unviable at the onset of freezing
Retention of CO2 in flash-frozen carbonated water is observed to be dependent on the temperature of the substrate during freezing. The state of CO2 retained in the resulting ices differs from clathrate hydrates, as inferred from the respective infrared spectra. Both mechanisms of CO2 retention are stable up to 140 K and under evacuated conditions.
In the context of Europa, the P–T states traversed by the samples plausibly represent the typical conditions around endogenous CO2, if it is indeed transported from the subsurface ocean to the surface while being retained in ice/frozen brines and/or liquid emerging on the surface. However, the absorptions of CO2 in laboratory infrared spectra do not match those detected on the leading side of Europa by the NIRSpec instrument on board JWST.
Therefore, it is unlikely that the endogenous CO2 observed at the surface of Europa is sourced directly from the ocean, unless additional processes affect the observed bands of CO2 on Europa.
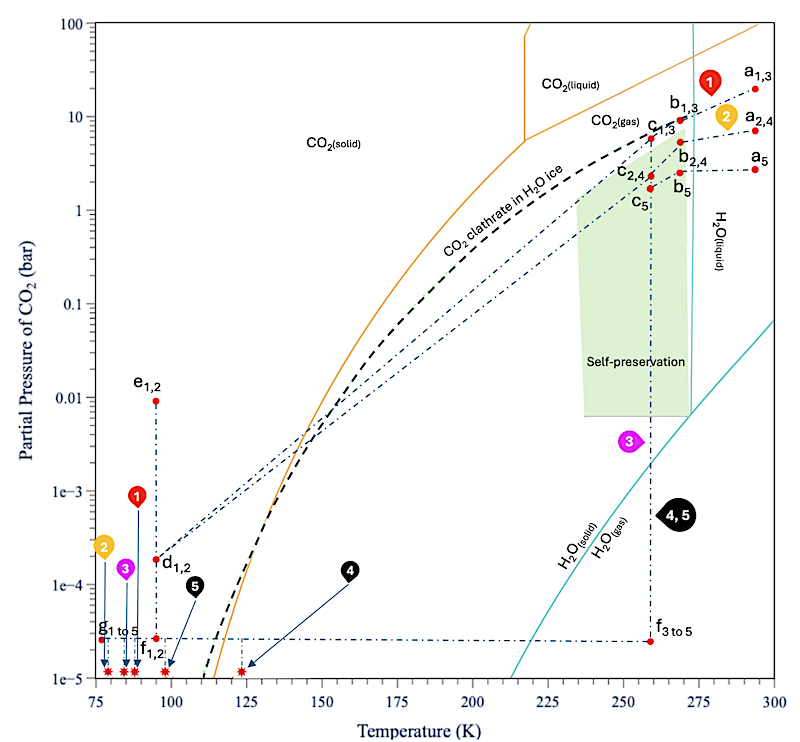
Pressure–temperature (P–T) conditions of our experiments compared to phase diagram of the CO2–H2O system. Red dots correspond to actual P–T conditions at various stages of ice preparation. Dashed lines are only indicative and do not correspond to measured variations in pressure. Paths labeled in red, yellow, and magenta correspond to ice retaining CO2. Paths labeled in black correspond to ice devoid of CO2. Lowercase letters show the stages of the experiment described in the text, with subscripts showing the applicable experiment number. The stars near the bottom left denote the P–T conditions during acquisition of spectra. — The Planetary Science Journal
The Fate of Frozen Carbonated Water at Europa-like Conditions, The Planetary Science Journal (open access)
Astrobiology,
Stay Informed With the Latest & Most Important News
-
 01Two Black Holes Observed Circling Each Other for the First Time
01Two Black Holes Observed Circling Each Other for the First Time -
 02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life
02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life -
 03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series)
03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series) -
 04True Anomaly hires former York Space executive as chief operating officer
04True Anomaly hires former York Space executive as chief operating officer -
 05Φsat-2 begins science phase for AI Earth images
05Φsat-2 begins science phase for AI Earth images -
 06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters
06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters -
 07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly
07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly