Now Reading: Mineral-facilitated Aqueous Synthesis Of Hydrogen Cyanide From Prebiotically Abundant Amino Acids For Chemical Evolution
-
01
Mineral-facilitated Aqueous Synthesis Of Hydrogen Cyanide From Prebiotically Abundant Amino Acids For Chemical Evolution
Mineral-facilitated Aqueous Synthesis Of Hydrogen Cyanide From Prebiotically Abundant Amino Acids For Chemical Evolution
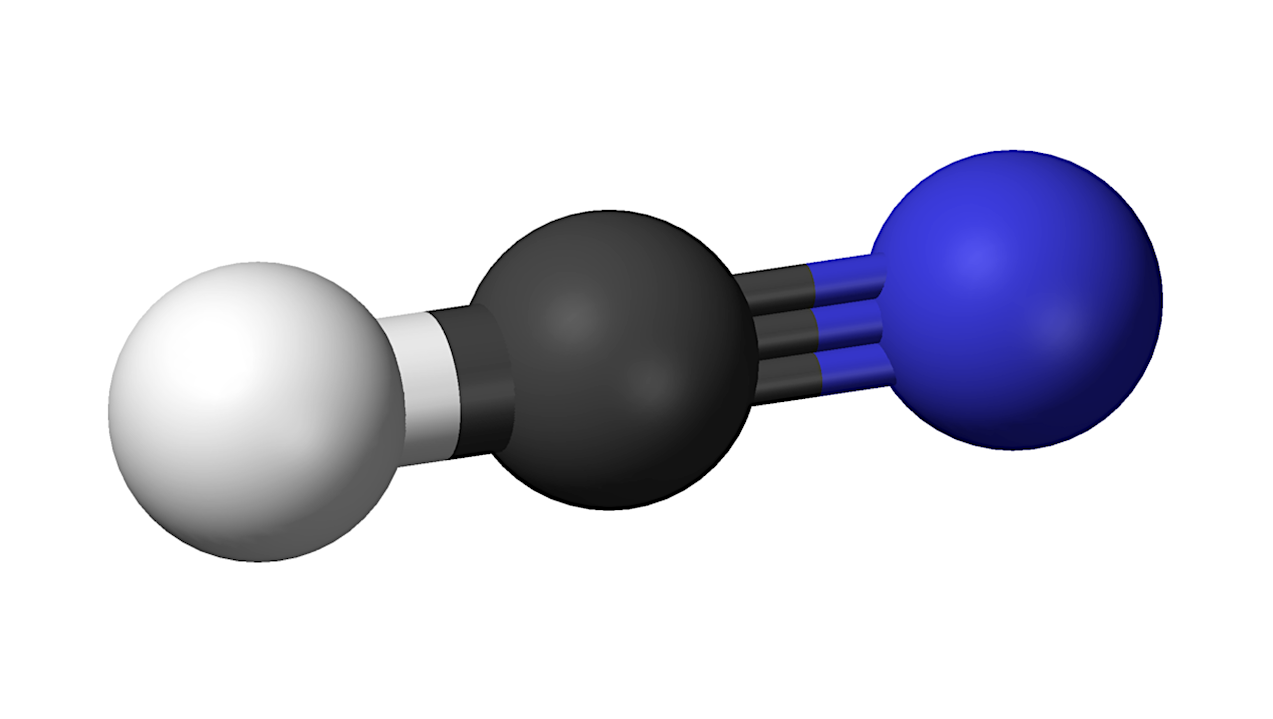
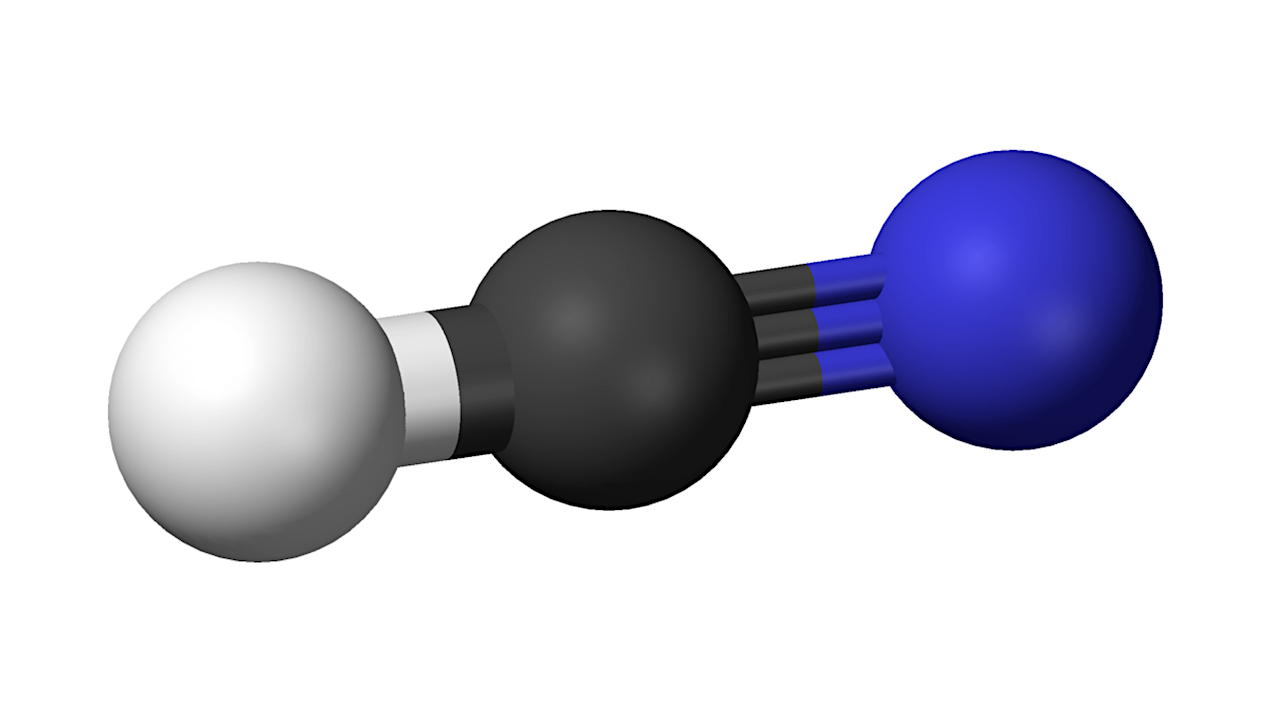
Hydrogen cyanide – Wikipedia
Recent advances in prebiotic chemistry suggest that hydrogen cyanide (HCN) serves as a fundamental precursor for nearly all essential biomolecules and protometabolic processes for life’s emergence.
Although prevailing models of endogenous cyanide production rely on atmospheric methane, the availability of methane remains uncertain. Unlike methane, amino acids were likely prebiotically abundant, forming through multiple synthetic pathways under a methane-free, nonreducing atmosphere.
Here, we demonstrate that HCN can form from proteinogenic amino acids in anaerobic aqueous solutions facilitated by geochemically available minerals, offering a route to overcome HCN scarcity under a nonreducing atmosphere. On manganese dioxide, the glycine-to-cyanide conversion proceeded across a broad range of pHs (2.0 to 12.6) and substrate concentrations (1 μM to 100 mM), achieving a maximum selectivity of 57%.
The reaction involves α-proton abstraction in amino acids, which is distinct from conventional chemical decarboxylation processes of amino acids, highlighting the unique role of MnO2 in activating the α-C-H bond to form HCN. HCN generation was observed for nearly all proteinogenic amino acids and short peptides.
Because amino acids can be synthesized from HCN-independent pathways from abundant carbon sources such as CO2 and CO, amino acid-derived HCN alleviates the requirement on atmospheric methane and can sustain continued chemical evolution in ambient aqueous environments.
Astrobiology, Astrochemistry,
Stay Informed With the Latest & Most Important News
Previous Post
Next Post
-
 01Two Black Holes Observed Circling Each Other for the First Time
01Two Black Holes Observed Circling Each Other for the First Time -
 02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life
02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life -
 03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series)
03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series) -
 04True Anomaly hires former York Space executive as chief operating officer
04True Anomaly hires former York Space executive as chief operating officer -
 05Φsat-2 begins science phase for AI Earth images
05Φsat-2 begins science phase for AI Earth images -
 06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters
06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters -
 07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly
07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly