Now Reading: The Formation of Glycolamide on Interstellar Ices
-
01
The Formation of Glycolamide on Interstellar Ices
The Formation of Glycolamide on Interstellar Ices

Graphical Abstract — ACS Central Science
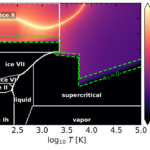
For decades, scientists have searched interstellar space for the chemical seeds of life. In this issue of ACS Central Science, Kaiser and co-workers (1) offer a striking advance in that search by demonstrating for the first time how glycolamide, a molecule closely related to amino acids, can form efficiently under the harsh and cold conditions of the interstellar medium (ISM).
Despite decades of effort, glycine, the simplest amino acid and a fundamental building block of proteins, has remained undetected in space. This absence has puzzled astrochemists because glycine has been detected in meteorites, comets, and even asteroid samples returned to Earth. (2−5)
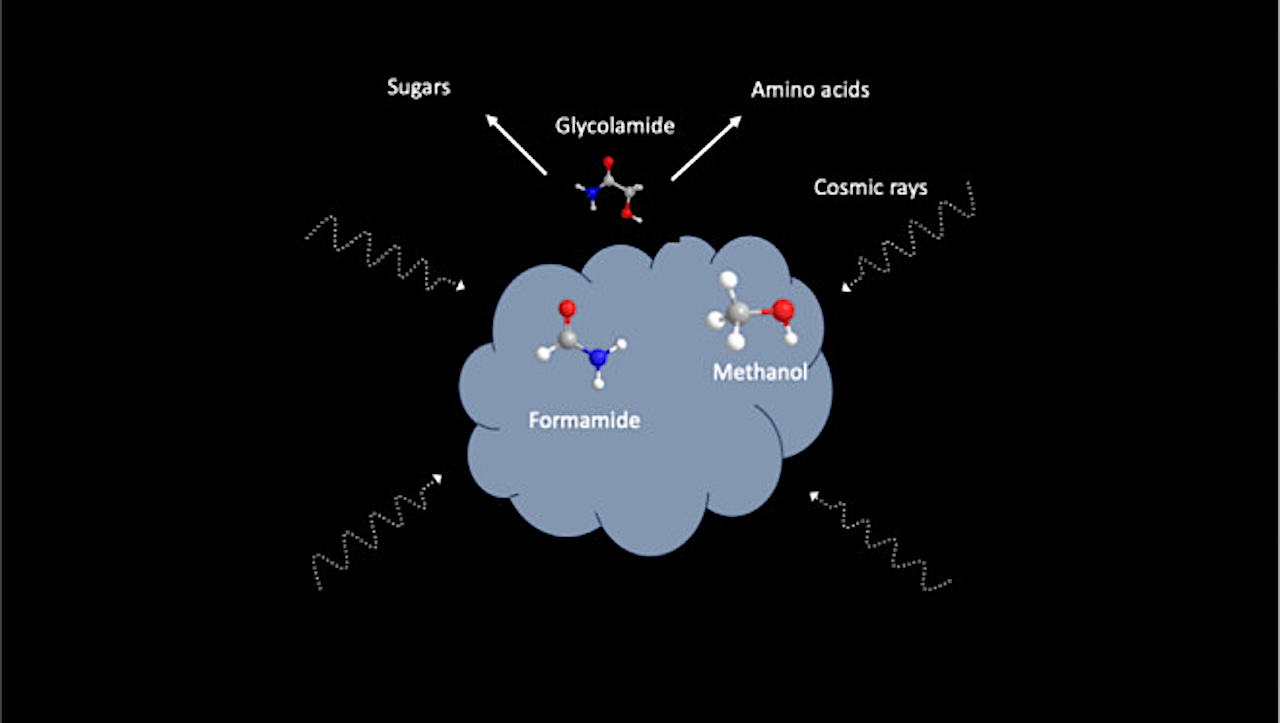
This paradox sets the stage for Kaiser’s research. Glycolamide is a small organic molecule that has recently been detected in the ISM. (6) While glycolamide is not itself an amino acid, it is in fact structurally related to glycine. The authors outline a chemical family tree where glycolamide serves as a parent to even more biologically significant molecules. Through various reactions, it can transform into glycine (the missing amino acid), glycolic acid, and other compounds essential for life.
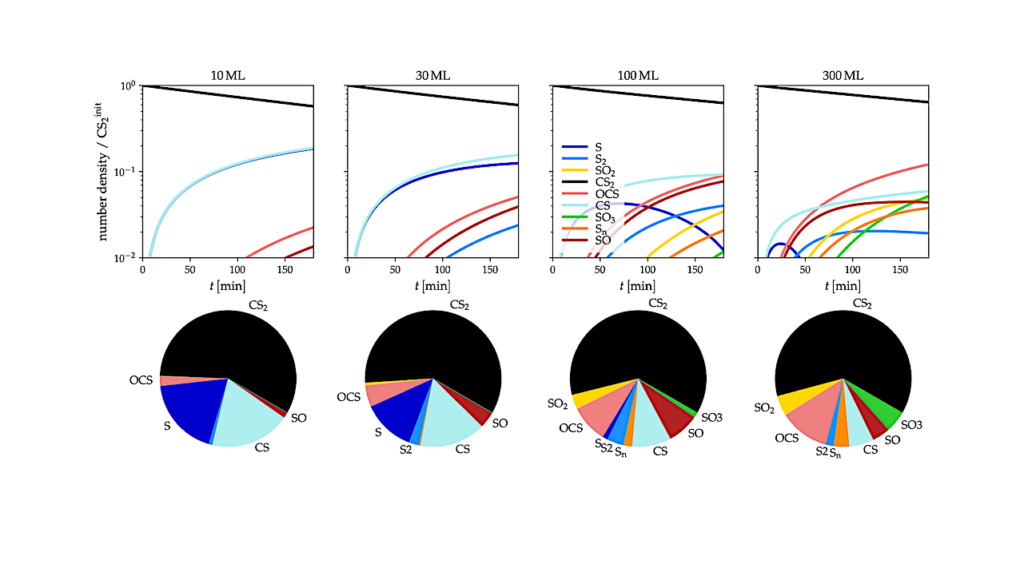
This implies that the “missing” glycine might simply be locked away in ices or constantly evolving from precursors like glycolamide. Kaiser and co-workers’ study focuses on the chemical pathways that might lead to glycolamide. They tackle the investigation by combining experimental and theoretical approaches. In the laboratory they recreate key features of the ISM.
They deposited thin icy films of simple, well-known interstellar molecules─formamide and methanol─onto cold surfaces of just a few degrees above absolute zero. These ices were then irradiated with energetic electrons that mimic the secondary particles produced by galactic cosmic rays. The authors performed theoretical calculations to help interpret their experimental results. Over time, the experiments revealed that glycolamide forms efficiently through radical chemistry inside these ices.
Their finding is important because it challenges an intuitive assumption about chemistry: that complex molecules require warmth, liquid solvents, or long reaction times. In contrast, the authors show that chemistry in space can be relatively fast, efficient, and surprisingly productive, even at temperatures near 5 K.
Astrobiology, Astrochemistry, interstellar,
Stay Informed With the Latest & Most Important News
Previous Post
Next Post
-
 01Two Black Holes Observed Circling Each Other for the First Time
01Two Black Holes Observed Circling Each Other for the First Time -
 02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life
02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life -
 03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series)
03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series) -
 04True Anomaly hires former York Space executive as chief operating officer
04True Anomaly hires former York Space executive as chief operating officer -
 05Φsat-2 begins science phase for AI Earth images
05Φsat-2 begins science phase for AI Earth images -
 06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters
06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters -
 07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly
07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly