Now Reading: Novel Chemical Pathways For The Formation Of Nucleobase Precursors Via Benzene π-Bond Addition To HCN
-
01
Novel Chemical Pathways For The Formation Of Nucleobase Precursors Via Benzene π-Bond Addition To HCN
Novel Chemical Pathways For The Formation Of Nucleobase Precursors Via Benzene π-Bond Addition To HCN
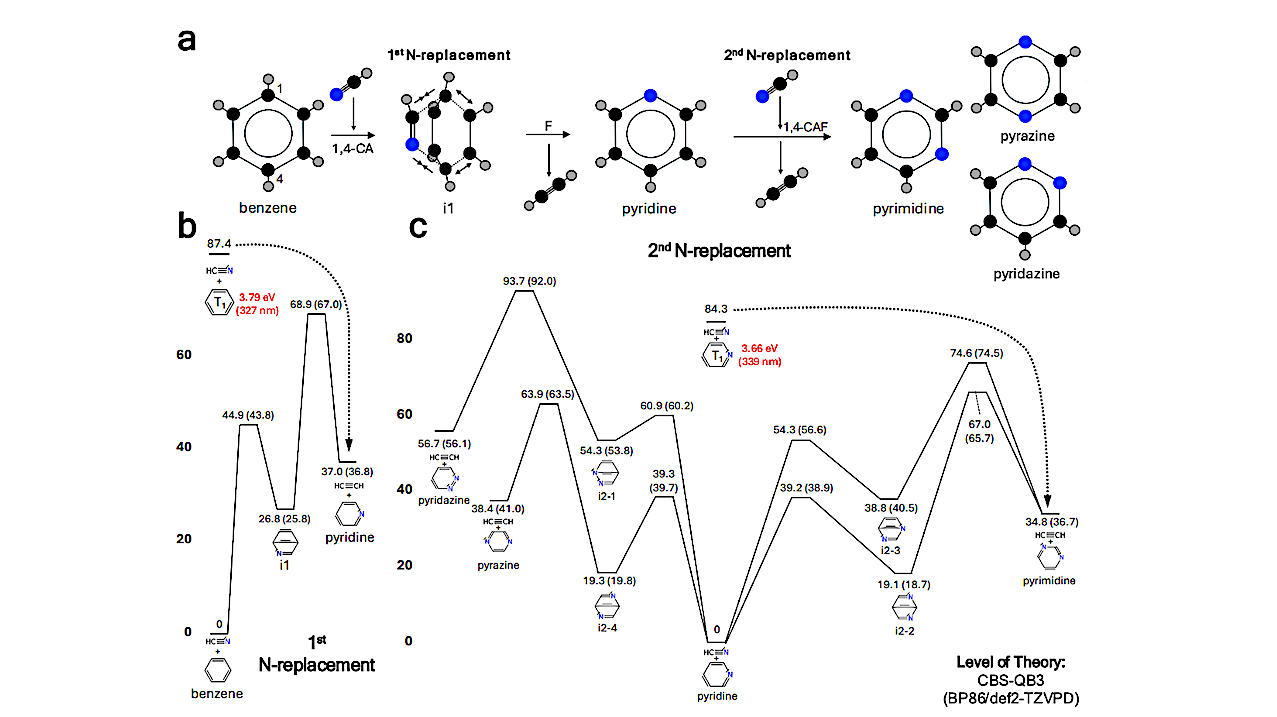
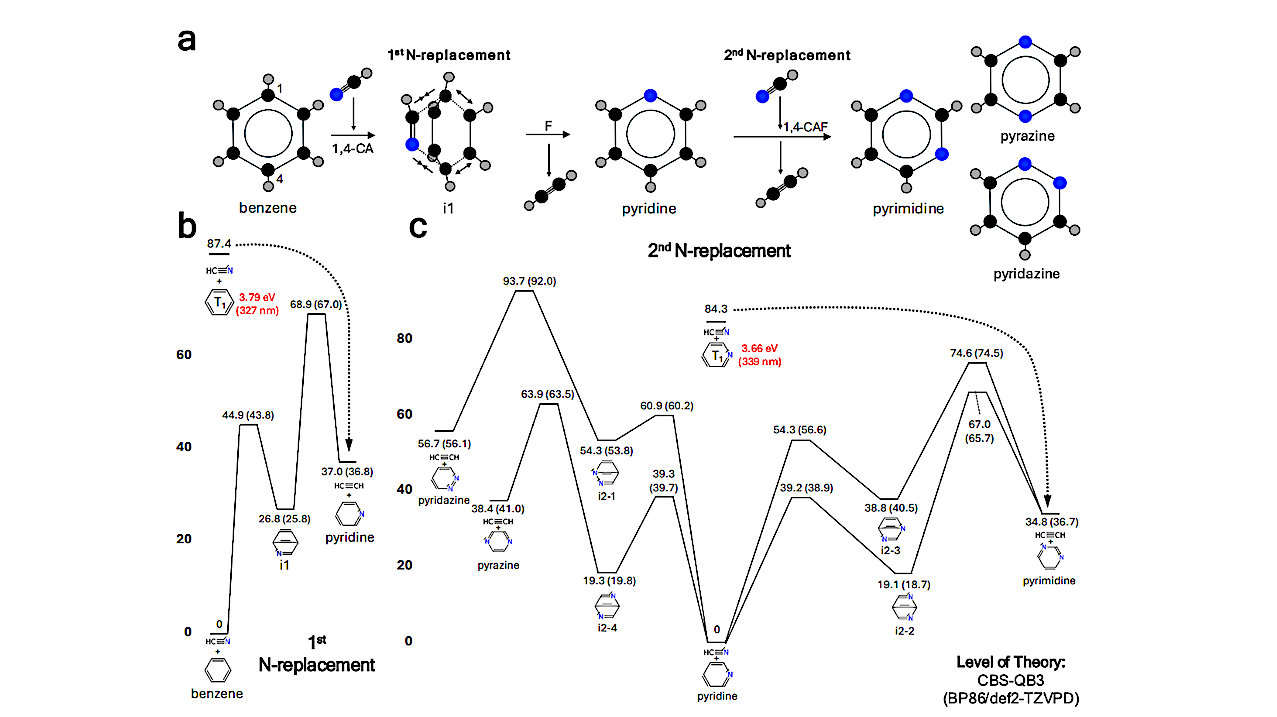
Overall information on the first and second nitrogen replacements in a benzene ring. a. The schematic diagram illustrating the formation of C4H4N2 isomers (pyrimidine, pyrazine, and pyridazine) via 1,4-cycloaddition and fragmentation (CAF) from the benzene + HCN reaction. Each colored ball represents a different atomic species: black for carbon, gray for hydrogen, and blue for nitrogen. CA stands for cycloaddition, and F stands for fragmentation. b. The C7H7N potential energy surface (first nitrogen replacement). c. The C6H6N2 potential energy surface (second nitrogen replacement). Numbers indicate potential energy (units in kcal/mol) calculated at the CBS-QB3 level of theory, while numbers in parentheses indicate potential energy calculated at the BP86/def2-TZVPD level of theory, considering water solvation effect at 298 K. The red numbers indicate the equivalent energy (in eV) and the corresponding wavelength (in nm) of the energy values shown in kcal/mol (black numbers) for each photoexcited triplet state (T1) of benzene and pyridine. The black dotted lines indicate well-skipping reactions which lead to the formation of pyridine+C2H2 and pyrimidine+C2H2, respectively. — physics.chem-ph
We propose a simple and efficient pathway for the formation of precursors to core nucleobases in DNA and RNA using a suite of computational chemistry methods.
Benzene, which is thermochemically stable in N2– or CO2-dominated atmospheres, could have formed via upper-atmospheric photochemistry or surface lightning and accumulated on the early Earth or Mars. However, nitrogen insertion into the benzene ring to form pyrimidine and purine is widely considered to be challenging.
We propose that nitrogen incorporation occurred through HCN 1,4-cycloaddition to benzene’s pi-system, followed by a C2H2 fragmentation mechanism, as confirmed by quantum chemistry calculations. This pathway, potentially facilitated by photochemistry at the ocean surface or episodic impact events on local reservoirs, can lead to pyrimidine formation, which can further react with NH3 and HCN to produce purine.
Extending this pathway to early Mars, our photochemical model simulates heterocyclic compound formation under cold, dry surface conditions that favor high benzene and HCN concentrations but lack liquid water.
We thus propose that organics formed during dry phases may have later dissolved into surface waters during wet phases and become concentrated as ocean sediments. This result supports Mars Sample Return efforts focused on ancient aqueous environments likely to retain prebiotic signatures.
Jeehyun Yang, Danica J. Adams, Renyu Hu, Yuk L. Yung
Comments: 16 pages, 6 figures, accepted for publication in Icarus Special issue entitled ‘Carbon in Planetary Environments: Sources and Evolution.’ We dedicate this paper to the memory of Professor Yuk L. Yung (1946-2026), who passed away on March 16, 2026
Subjects: Chemical Physics (physics.chem-ph); Earth and Planetary Astrophysics (astro-ph.EP)
Cite as: arXiv:2605.00035 [physics.chem-ph] (or arXiv:2605.00035v1 [physics.chem-ph] for this version)
https://doi.org/10.48550/arXiv.2605.00035
Focus to learn more
Submission history
From: Jeehyun Yang
[v1] Tue, 28 Apr 2026 06:08:37 UTC (849 KB)
https://arxiv.org/abs/2605.00035
Astrobiology, Astrochemistry,
Stay Informed With the Latest & Most Important News
Previous Post
Next Post
-
 01Two Black Holes Observed Circling Each Other for the First Time
01Two Black Holes Observed Circling Each Other for the First Time -
 02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life
02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life -
 03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series)
03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series) -
 04True Anomaly hires former York Space executive as chief operating officer
04True Anomaly hires former York Space executive as chief operating officer -
 05Φsat-2 begins science phase for AI Earth images
05Φsat-2 begins science phase for AI Earth images -
 06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters
06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters -
 07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly
07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly