Now Reading: Methanimine As A Sink In The HCN And HNC Solid State Hydrogenation Network
-
01
Methanimine As A Sink In The HCN And HNC Solid State Hydrogenation Network
Methanimine As A Sink In The HCN And HNC Solid State Hydrogenation Network
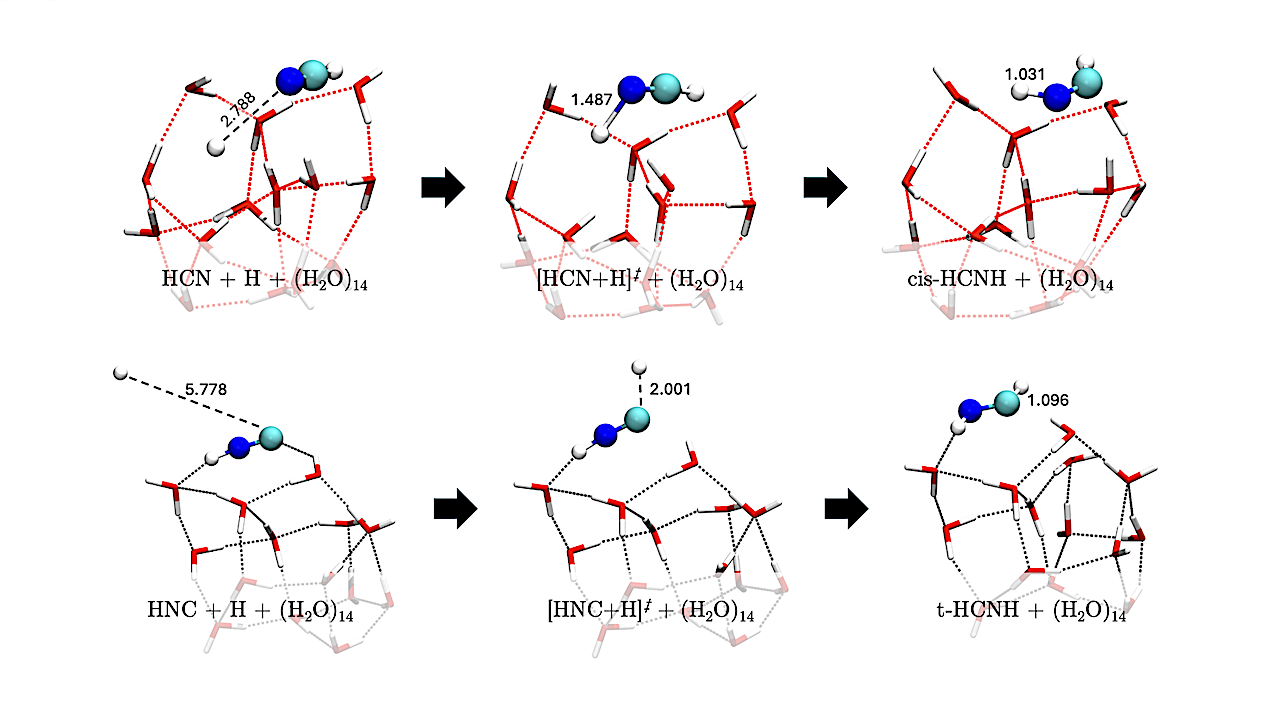
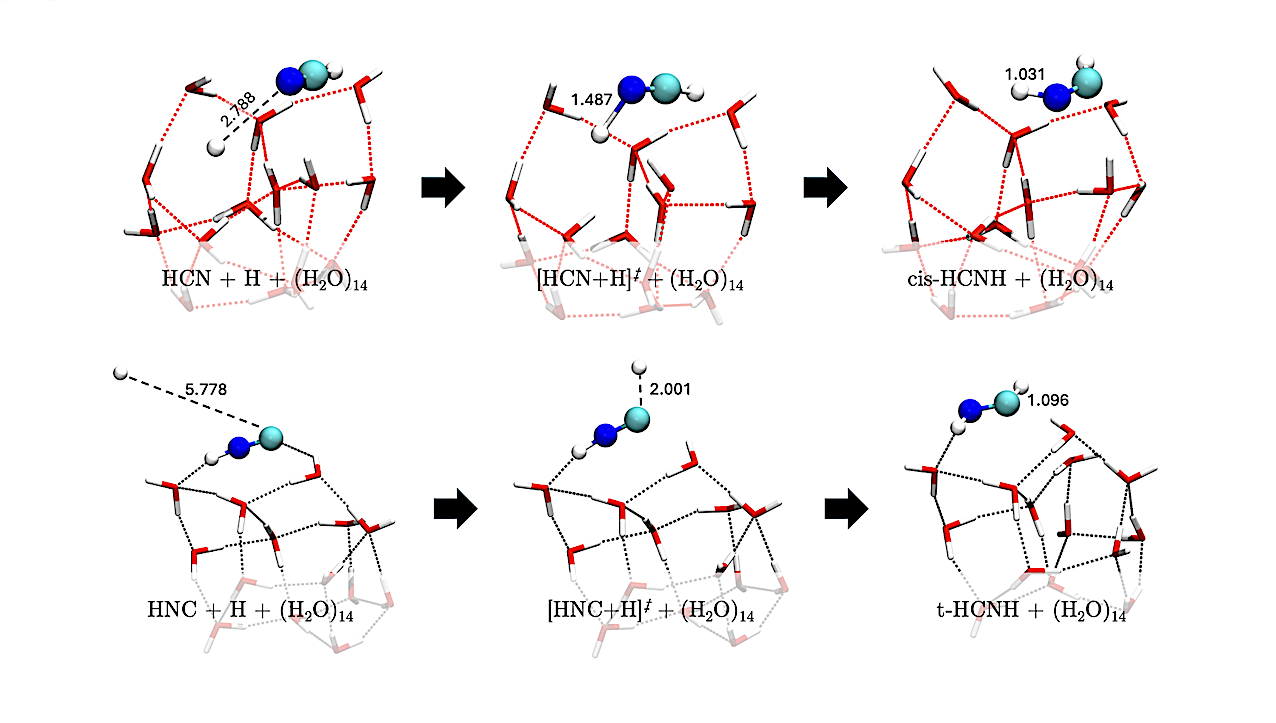
Geometries of the H-addition reactions on HNC leading to cis- and trans-HC•NH (reactions R4-cis and R4-trans). Distances are in Å — astro-ph.GA
We aim to provide a systematic and quantitative description of the hydrogenation network connecting HCN and HNC to methylamine on interstellar water ices, while identifying dominant pathways and bottlenecks.
To this end, we performed a comprehensive quantum-chemical investigation of H-addition, H-abstraction, reactions with H2, and water-assisted H-transfer isomerization, covering intermediates linking HCN and HNC to CH3NH2. Calculations were carried out on amorphous solid water clusters of 14 molecules.
Using benchmarked density functional theory, we derived activation barriers, elucidated mechanisms, and determined the binding energy distribution of H2CN and CNH2, also assessing deuterium substitution effects. H-addition reactions generally involve activation barriers, except for radical species.
Considering both barrier heights and tunneling crossover temperatures, the most favorable sequence originates from HNC rather than HCN. The network evolves toward methanimine (H2CNH), the central species, or the singlet carbene HC:NH2, from which further hydrogenation leads to methylamine. Along these paths, several reactions are barrierless, while some H-abstraction processes compete with addition. Reactions involving H2 are uncommon, as most are endoergic.
Deuterium substitution weakly affects classical barriers but significantly influences tunneling efficiencies. Our results support efficient formation of methanimine and methylamine from HNC on cold interstellar ices, with methanimine acting as a chemical sink, whereas HCN is less reactive and more likely to persist. These findings provide quantitative constraints for astrochemical models.
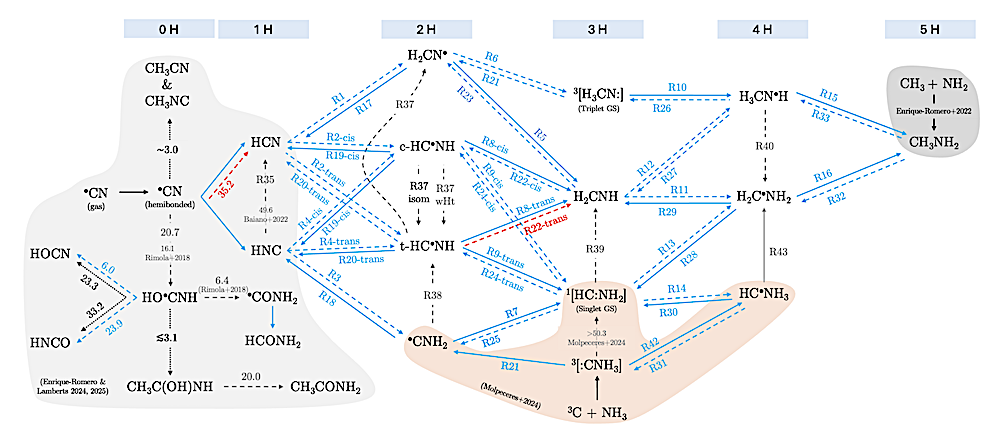
Schematic representation of the •CN chemical network on interstellar ice surfaces. The figure summarizes the reactions investigated in this work (the hydrogenation of HCN and HNC) together with related processes reported in previous studies (Enrique-Romero et al. 2022; Molpeceres et al. 2024; Enrique-Romero & Lamberts 2024; Enrique-Romero & Lamberts 2025b), shown as colored regions. Unless otherwise indicated, radical species have doublet spin multiplicity. Solid arrows denote barrier-less reactions, whereas dashed arrows indicate reactions with an activation barrier. Activation energies are given in kJ mol−1 . Arrow colors indicate reaction types: blue for H addition and abstraction, red for reactions with H2, gray for wHts, and black for other processes (e.g., isomerization or radical–radical reactions). Energies are reported on a per-reaction basis, assuming efficient thermalization on amorphous solid water, such that each activation barrier is referenced to the products of the preceding step. — astro-ph.GA
Joan Enrique-Romero, Thanja Lamberts
Comments: Accepted in A&A (Apr 2026)
Subjects: Astrophysics of Galaxies (astro-ph.GA)
Cite as: arXiv:2604.09228 [astro-ph.GA](or arXiv:2604.09228v1 [astro-ph.GA] for this version)
https://doi.org/10.48550/arXiv.2604.09228
Focus to learn more
Submission history
From: Joan Enrique-Romero
[v1] Fri, 10 Apr 2026 11:36:56 UTC (5,340 KB)
https://arxiv.org/abs/2604.09228
Astrobiology, Astrochemistry,
Stay Informed With the Latest & Most Important News
Previous Post
Next Post
-
 01Two Black Holes Observed Circling Each Other for the First Time
01Two Black Holes Observed Circling Each Other for the First Time -
 02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life
02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life -
 03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series)
03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series) -
 04True Anomaly hires former York Space executive as chief operating officer
04True Anomaly hires former York Space executive as chief operating officer -
 05Φsat-2 begins science phase for AI Earth images
05Φsat-2 begins science phase for AI Earth images -
 06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters
06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters -
 07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly
07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly