Now Reading: How Rock Removes CO2 From The Atmosphere
-
01
How Rock Removes CO2 From The Atmosphere
How Rock Removes CO2 From The Atmosphere

Graphical Abstract — ACS Nano
Rocks can bind carbon dioxide — and much faster than previously thought. For a long time, it was assumed that the transformation of CO2 into carbonate rock depends on very slow, time-consuming processes. According to that view, the binding of CO2 injected industrially into the ground would take centuries.
However, practical observations and theoretical calculations suggested that there may also be a much faster route from CO2 to carbonate, mediated by water acting somewhat like a catalyst. This suspected mechanism has now been demonstrated for the first time at TU Wien, using imaging techniques on the atomic scale.
A long-known process that takes centuries
How can carbon dioxide turn into rock? For a long time, it was thought that this requires two steps: first, CO2 has to dissolve in water and form charged particles; second, the rock itself (for example silicates in the ground) must partially dissolve. A new material can then form, permanently incorporating the carbon from the carbon dioxide into the rock.
“However, this is a very sluggish process,” says Giada Franceschi, who led the project together with Prof. Ulrike Diebold at the Institute of Applied Physics at TU Wien. “It cannot explain why this kind of carbonate rock often forms very quickly in nature. Tests involving industrial CO2 injection into the ground show that 60% of the carbon can already be bound in minerals within two years. If ions first had to dissolve out of the rock, that would take decades or centuries.”
A direct pathway, demonstrated for the first time
For quite some time, however, researchers had speculated that there might be a more direct way to incorporate carbon dioxide into certain materials: in the presence of water molecules on the mineral surface, CO2 might be incorporated directly into the rock, without the mineral having to dissolve first and without taking the detour through dissolved ions, whose formation is chemically rather slow. And under natural conditions, water is almost always present around such minerals.
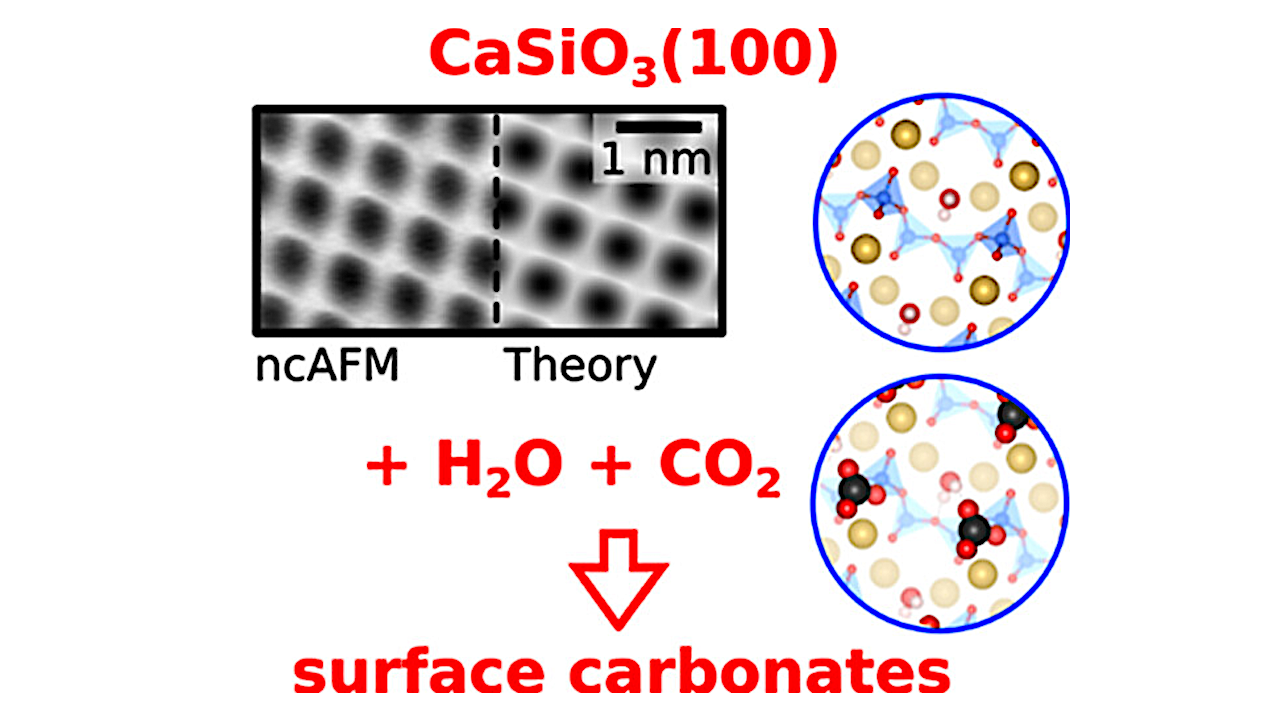
Using the mineral wollastonite, the team at TU Wien has now shown that this alternative pathway really exists. This was made possible by high-resolution atomic force microscopy: the chemical processes could be observed directly on the atomic scale.
The CO2 molecule has to bend
“If there is a thin layer of water on the wollastonite surface, its interaction with carbon dioxide changes in a decisive way,” explains Ulrike Diebold. “From a geometrical point of view, carbon dioxide is normally completely straight. The two oxygen atoms bonded to the carbon point in exactly opposite directions. But water on the wollastonite surface can bend the carbon dioxide molecule — and that changes its chemical properties.”
The “bent” carbon dioxide molecule can then attach directly to the wollastonite, forming a stable bond — without the wollastonite having to dissolve first. “Without water, this is not possible, because the right docking site is missing,” says Giada Franceschi. “But even a tiny amount of water is enough to completely change the interaction between CO2 and wollastonite.”
This is the first direct demonstration of the crucial mechanism that enables rapid CO2 capture not only in wollastonite, but most likely also in other similar minerals. “If, in the future, we want to remove CO2 from the atmosphere and store it permanently for unlimited periods of time, then we need to turn it into solid rock,” says Ulrike Diebold. “Our measurements show which effects on the atomic scale can be used to achieve that.”
Molecular Views of Mineral Carbonation: Reaction of CO2 with the Wollastonite (100) Surface, ACS Nano (open access)
Astrobiology, Astrogeology,
Stay Informed With the Latest & Most Important News
Previous Post
Next Post
-
 01Two Black Holes Observed Circling Each Other for the First Time
01Two Black Holes Observed Circling Each Other for the First Time -
 02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life
02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life -
 03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series)
03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series) -
 04True Anomaly hires former York Space executive as chief operating officer
04True Anomaly hires former York Space executive as chief operating officer -
 05Φsat-2 begins science phase for AI Earth images
05Φsat-2 begins science phase for AI Earth images -
 06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters
06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters -
 07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly
07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly