Now Reading: The Master Molecule That Built Biology: How Water Shaped The Chemistry Of Life
-
01
The Master Molecule That Built Biology: How Water Shaped The Chemistry Of Life
The Master Molecule That Built Biology: How Water Shaped The Chemistry Of Life
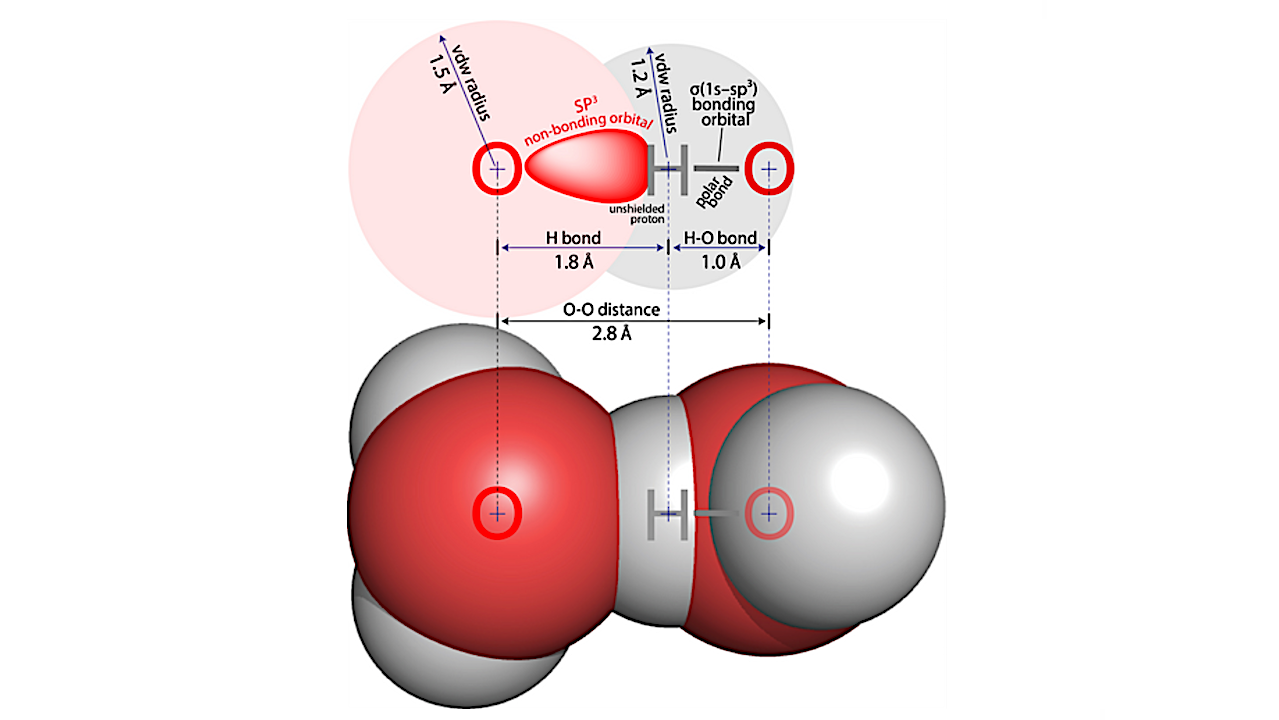
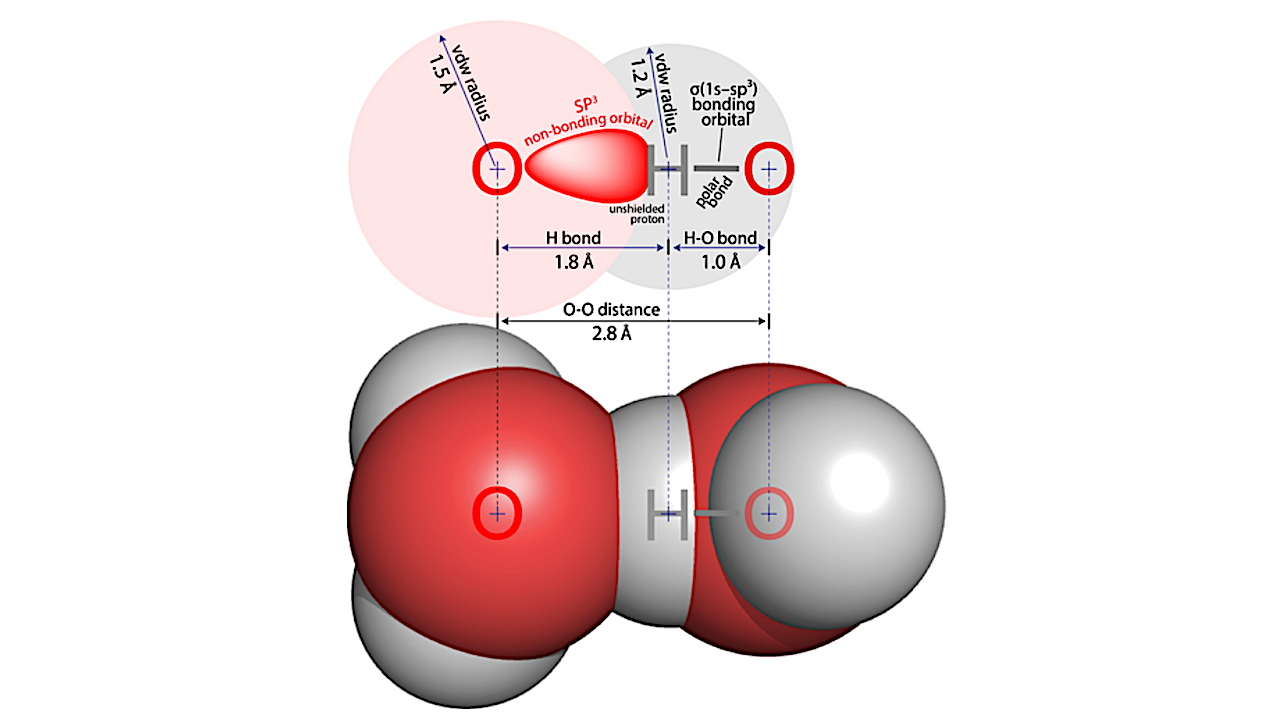
In a water–water hydrogen bond, a hydrogen nucleus from one water molecule is donated to an sp3 lone pair on the oxygen atom of a neighboring water molecule, along the axis of the O–H covalent bond. The polar O–H bond withdraws electron density from the hydrogen atom, exposing the back of the proton to HB acceptor. — Protein Science via PubMed
The deep entanglement of biomolecular structure and function with aqueous systems supports the view that water actively sculpted both molecules and processes during the origins of life and continues to constrain evolution today.
Nature’s rules of biochemistry and biophysics have survived for nearly 4 billion years. The in vivo roles of water, the architectures of biopolymer backbones and side chains, the structure and function of ribose, ATP, the translation system, the genetic code, and certain inorganic cations persist unchanged.
Hydrogen bonding and the hydrophobic effect predate life on Earth altogether. Here we examine fundamental forces that established, shaped, and continue to constrain biochemistry and biophysics.
The results support a model in which life emerged through water‐based selection among diverse molecules and molecular ensembles, with molecular fitness defined by behaviors in and interactions with water.
Life is thus composed of molecules that cooperate with, resist, and exploit the unique properties of water. Biological molecules employ chemical strategies that enable selective and controlled persistence in aqueous environments, a phenomenon we classify as recalcitrance.
Molecular assembly reduces conformational heterogeneity, constrains dynamics, and sterically excludes reactive agents, including water and hydrolytic enzymes. By this mechanism, lifetimes of folded proteins, structured RNAs, assembled phospholipids, and polysaccharides are mediated by their organizational states.
Astrobiology, Biochemistry, Astrochemistry,
Stay Informed With the Latest & Most Important News
Previous Post
Next Post
-
 01Two Black Holes Observed Circling Each Other for the First Time
01Two Black Holes Observed Circling Each Other for the First Time -
 02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life
02From Polymerization-Enabled Folding and Assembly to Chemical Evolution: Key Processes for Emergence of Functional Polymers in the Origin of Life -
 03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series)
03Astronomy 101: From the Sun and Moon to Wormholes and Warp Drive, Key Theories, Discoveries, and Facts about the Universe (The Adams 101 Series) -
 04True Anomaly hires former York Space executive as chief operating officer
04True Anomaly hires former York Space executive as chief operating officer -
 05Φsat-2 begins science phase for AI Earth images
05Φsat-2 begins science phase for AI Earth images -
 06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters
06Hurricane forecasters are losing 3 key satellites ahead of peak storm season − a meteorologist explains why it matters -
 07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly
07Binary star systems are complex astronomical objects − a new AI approach could pin down their properties quickly